Pharmaceutical manufacturer Zydus Cadila got the approval of DGCI (Drugs Controller General of India) for a medication ‘Virafin’ to treat moderate COVID-19 patients. ‘Virafin’ is the brand name of Zydus’s Pegylated Interferon alpha-2b. First marketed as Heberon Alfa R, the medication was developed in 1986 by the Cuban Center for Genetic Engineering and Biotechnology of Havana as a drug against viral hepatitis C and melanoma – a type of skin cancer. Since then, it has been used with other antiviral drugs or with surgical procedures in these diseases.
Zydus has announced this development after releasing the news of another indigenous vaccine being developed called ZyCoV-D, which has allegedly completed phase 1 and 2 trials and is expected to be released in 2022. Zydus Cadila, in its phase III clinical trial registry for Virafin, suggested it has shown statistically significant clinical improvements in phase II studies with COVID-19 patients.
Since Zydus is marketing the drug for moderate COVID-19 and not for severe disease, the primary endpoint is reducing hospital time and increasing the recovery rate instead of mortality. However, using published scientific research, we will evaluate the impact of Virafin on mortality and hospital time in COVID-19 patients. Additionally, we will discuss whether interferons can mechanistically work against COVID-19.
Claim:
Zydus-Cadila claims that Virafin might reduce recovery time or hospitalisation period during COVID-19.
Verdict:
False
Fact-Check:
What is Virafin?
Virafin (or PegIntron, Sylatron in the US) is not a direct antiviral but a biological response modulator. It is used as an antineoplastic agent (anti-cancer drug) and is administered subcutaneously (under the skin). This drug is a protein alpha- interferon-2b released from human cells and is known for its antiviral and anti-proliferative activity. This protein is one of the 16 alpha-interferon sub-categories, which have a high affinity for cell surface receptors. In Virafin, this protein ‘alpha- interferon-2b is attached to a polymer derived from petroleum to increase its therapeutic effect.
The Zydus clinical trial of Virafin
Zydus published a phase II clinical trial in March 2021 that evaluated the data for Virafin against 20 COVID-19 patients, and which was compared with Standard-of-Care (SOC) against a cohort of another 20 COVID-19 patients. After two doses of Virafin injection on day 1 and 8 in a hospital setting, only 1 out of 20 patients receiving Virafin was SARS-CoV-2 positive on day 14. In comparison, among those who did not receive Virafin (were on the SOC), 6 out of 20 patients were SARS-CoV-2 positive on day 14.
The number of participants in this study was deficient (20 in each group). Also, the study excluded all patients with comorbidities like kidney and liver disease and were all under the age of 65. Since most patients recover from COVID-19 on their own without external therapeutic intervention, it is impossible to show whether Virafin affected their recovery prospects, i.e., if they recovered on their own, especially in a tiny sample size of 20.
Also, it is impossible to ascertain the 1st day of infection when the patient is hospitalised after presenting with symptoms. When the patient is first infected, it takes about 4-6 days until the first signs of symptoms of Covid-19 are observed. The patient is likely 6-8 days or more into the infection when they present themselves at the hospital. Thus, the endpoints measured on day 14 cannot be a direct comparison between individuals. Such effects can only be resolved with large data sets, i.e., studies with a sample size of 1000 or more patients.
Secondly, the phase III data is not published, which is meant for a 250 patient cohort according to its clinical trial registry (CTRI). The CTRI also details the length of the trial, ten months from December 2020, which is estimated to be between October-November 2021. It is not clear when all the patients were enrolled and the actual study began. In the best case, even if the study enrolled all patients in January, one would just have data for just three months until April. Additionally, the patient cohort size for this Phase III study is also much smaller than what is statistically needed to identify any substantial therapeutic effect of the drug candidate. It is not possible to make a categorical claim about the drug candidate’s therapeutic efficacy with this small patient cohort.
Did Virafin reduce COVID-19 severity or recovery time in the Zydus phase II study?
The inflammatory response that arises as a result of interferon alpha-2 is also similar to the cytokine storm inflammation of the interferon protein family that occurs as a result of the COVID-19 infection. Such inflammation in severe COVID-19 is the reason for tissue damage, multiple organ dysfunction, and in some cases, death. Nonetheless, in mild-moderate disease, repair processes are initiated as soon as the inflammation is triggered. Thus, the body’s recovery system is engaged in healing post-inflammatory processes.
The question that needs to be answered is whether the data presented from the Phase II study, a study that screened out severe cases of Covid-19 showed no mortalities in both the drug candidate and the SOC group is due to the physiological action of the drug candidate, or a result of the body’s post-inflammatory healing process. Would the same effect be replicated in a large patient population, in both moderate and severe categories? Since the moderate disease patients recover independently without any external therapeutic intervention, it is important to control for this confounding factor during the study.
Virafin may have had little impact on the recovery of the moderate COVID-19 patients, as its effect is primarily related to evoking the immune response similar to the cytokine storm in detecting viral antigen. However, the small sample size of this Phase II study does not allow us to answer this question statistically.
Are interferons helpful or harmful for COVID-19 hospitalisation? 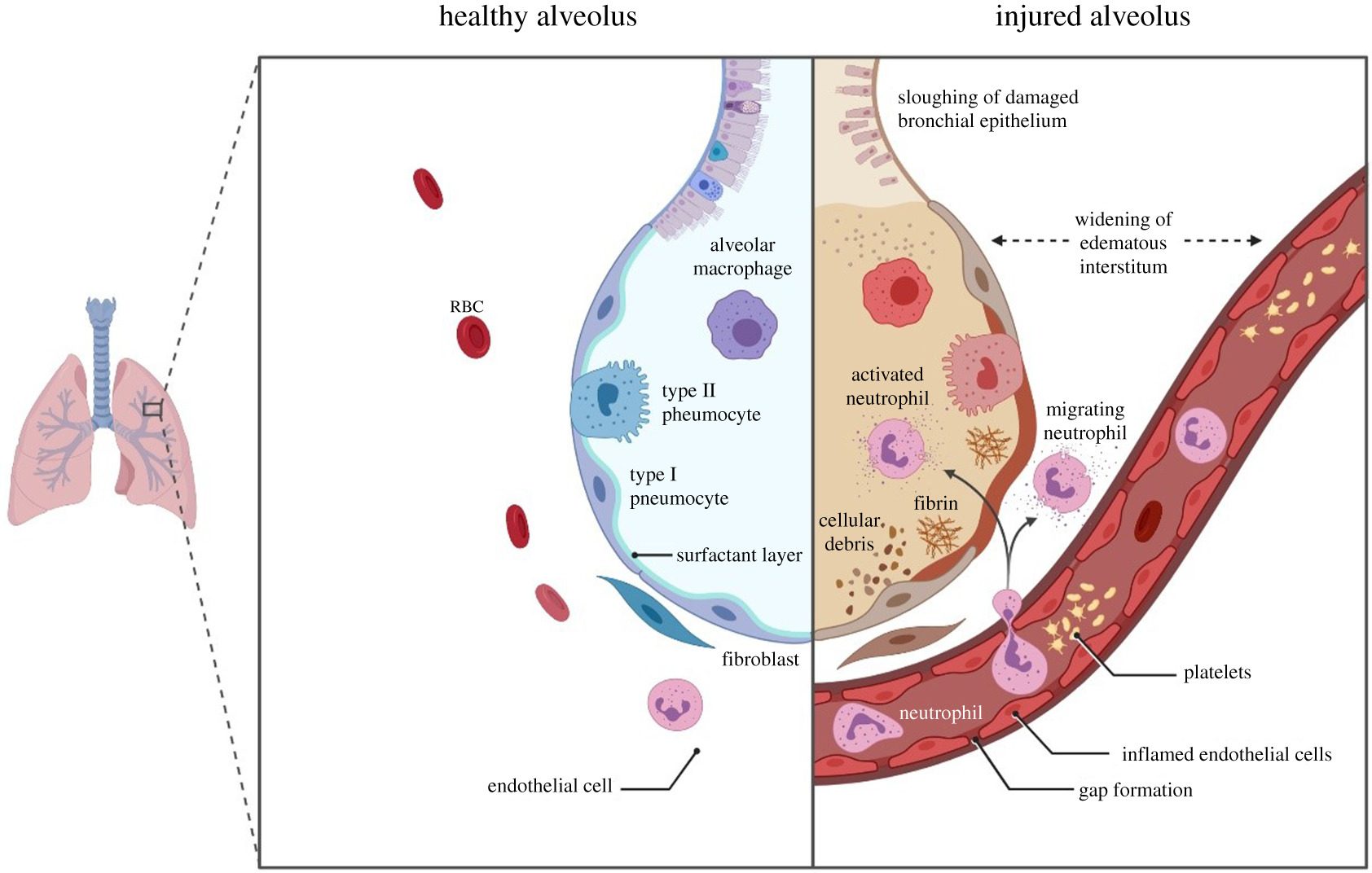
Much of the research related to SARS-CoV-2 in the early phase of the pandemic was focused on the mechanisms by which the disease caused deaths. It is now undisputed that the cytokine storm is linked with extensive lung damage and deterioration during COVID-19. Interferons are a type of central cytokines, and some of them, i.e. interferons- alpha, beta and gamma, are produced after a viral infection. These different interferons are produced by various immune cells to generate an immune response that results in inflammation, which also damages the organs. The figure above (Fara et al. 2020) suggests how various active immune cells in a hyper-inflammatory state can damage the healthy balloon-shaped alveoli of the lungs.
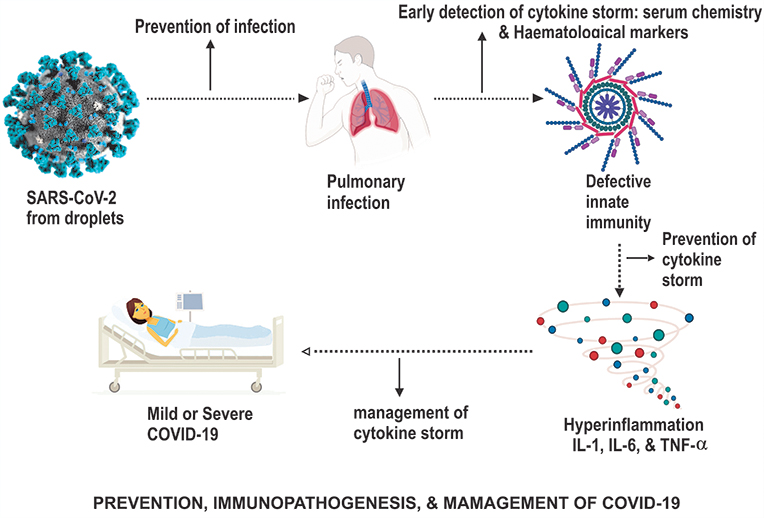
Further, the general line of treatment and management in COVID-19 has been about prevention and management of the cytokine storm, not invoking it. Adding to the cytokine storm, drugs may cause more harm. For example, an immune-suppressor steroidal drug, i.e. dexamethasone, prednisone & methylprednisolone, are used precisely to suppress this hyper inflammation (see figure above). A significant amount of research, including WHO, has recommended the use of steroids in attenuating cytokine-storms with excessive interferon activity for severe COVID-19.
What does data from other studies suggest about the efficacy of interferons?
The following studies were conducted to ascertain if there was a possibility of using the body’s immune response to treat COVID-19 that doesn’t negatively affect the organ systems within the body. We will evaluate all those studies systematically here:
This study published in May 2020 was one of the first studies to show the impact of interferon-alpha 2b against moderate COVID-19. However, the patients receiving interferon were only 53, which was compared to 24 that received the standard care. Most importantly, the patients were not randomised in each group, and the time of hospitalisation from the onset of the first symptoms was the largest in the standard-care group. Thus, comparing recovery time in the hospital, the late hospital admission in the control group vs the interferon group was the obvious confounding factor and rendered the study futile.
Another study tested the use of this drug was from China and recruited 111 patients for the treatment group and compared it with 12 patients of standard care. However, instead of measuring the recovery period or hospitalisation time, the study measured the viral clearance and oxygen requirement in these patients. The study showed no differences in the interferon-alpha 2b group as opposed to the standard care, indicating that in a slightly larger group, the slight chance effect of interferon therapy diminishes.
Thus, in the light of studies 1 and 2 above, there is no evidence for any clinical benefits such as a viral reduction in hospitalised COVID-19 patients. The data of reduced hospital period is frail and confounded with the unmatched timeline of hospitalisation in each patient.
Why do in-vivo studies for the same treatment show a different picture?
Although, mechanistically, the idea is based on the scientific rationale that interferon I (alpha and beta) response is suppressed during SARS-CoV-2. Also, that impaired or delayed interferon-alpha and beta responses add to the severity of the disease. However, the reason for efficacy within in-vivo preparations may be that the external dosing with interferon-alpha 2b manipulates a viral replication at the time it is possible.
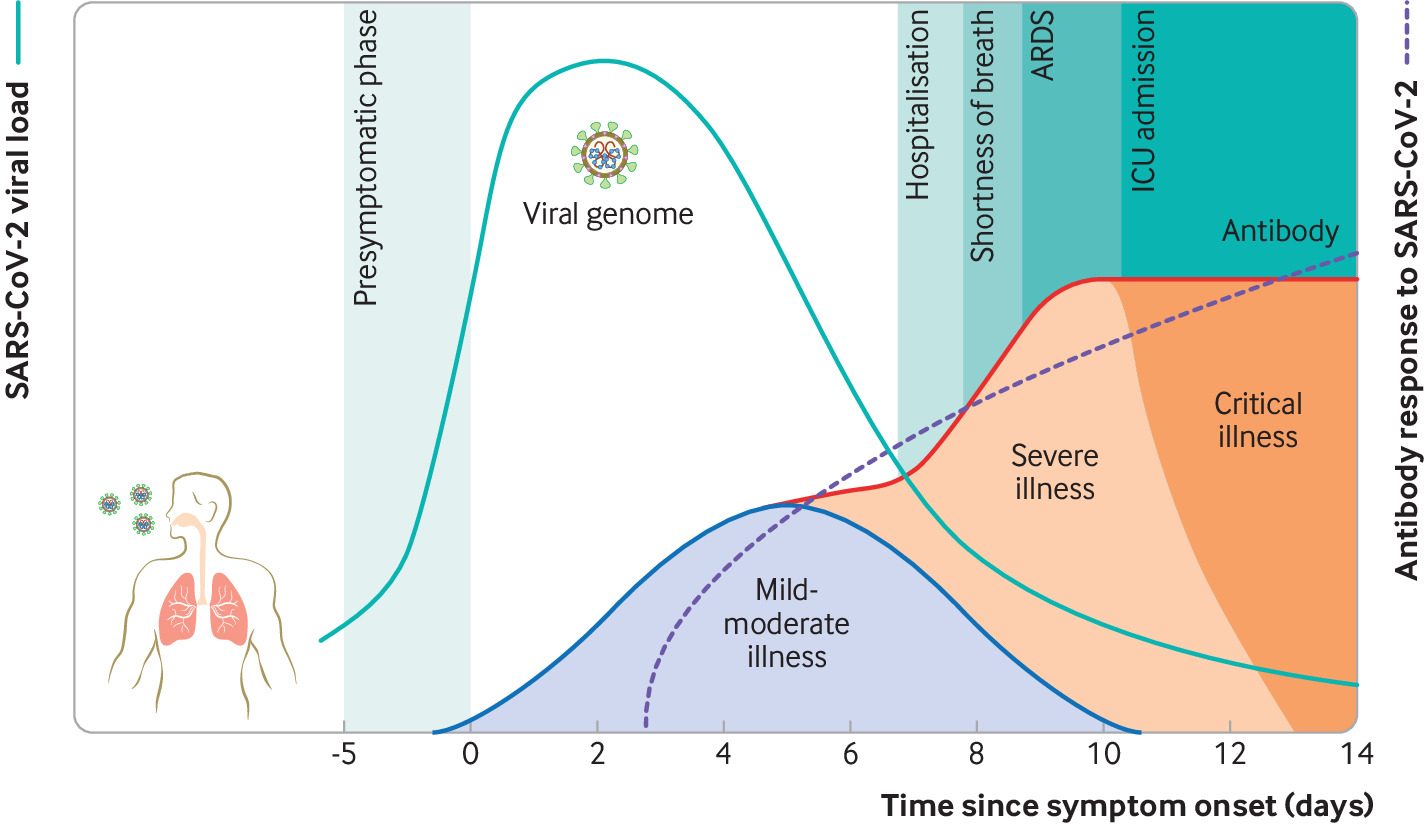
However, in humans, this replication time point is often much earlier in the disease history, i.e. when the symptoms are largely absent. Once the replication is over (1-6 days), the cytokine storm in moderate-to-severe cases produces respiratory symptoms and lung damage, leading to the patient’s hospitalisation (see picture above from Cevik et al. 2020). Thus, in the second half of the disease period, only suppression of the immune response via steroids, and other organ support, can help treat the damage by the cytokine storm. Administering antivirals or interferons after the viral genome replication period, i.e. during hospitalisation (days 6+), may not have any impact on the clinical symptoms.
Conclusion:
In the race to develop COVID-19 treatments, many drugs and vaccines have been approved at a faster rate, often skipping studying the therapeutic benefits in a well designed and controlled phase III study. In this case, interferon-alpha 2b, a repurposed drug from other diseases, has been approved by the Drug Controller General of India (DCGI) without robust clinical data.
The DCGI has used the ‘accelerated approval’ procedure in the Second Schedule of the New Drugs & Clinical Trials Rules, 2019 for this approval. To secure approval under this pathway, pharmaceutical companies have to show that their experimental drug candidate demonstrated ‘remarkable efficacy’ during the Phase II trials for a disease that lacks alternative treatments and that there is a prima facie case of meaningful therapeutic benefit over the existing treatments. Neither of these requirements seems to have been met with this study. Given the small patient cohort for this study, it would be important to understand how the DCGI determined that Virafin demonstrated “remarkable efficacy” in this 20+20 patient study. Furthermore, even the projected sample size for Phase III as documented in the CTRI is not large enough to clearly answer this question.
When most of the moderately ill patients infected with Covid-19 recover automatically from the disease without any external therapeutic intervention and that hospitalisation of patients occurs largely after the symptoms appear- which is also the period when the viral replication is near an end, it is not clear on what basis did the DCGI approve this drug for the treatment of this disease. We have reached out to Zydus multiple times for comment but are yet to get a response.
Independent journalism that speaks truth to power and is free of corporate and political control is possible only when people start contributing towards the same. Please consider donating towards this endeavour to fight fake news and misinformation.